Every atom forms the building blocks of all matter, making atomic structure one of the most fundamental concepts in Chemistry.
The periodic table organises all chemical elements based on their atomic number and mass number, allowing scientists to predict chemical properties and reactions.
The atomic structure of an element determines how it behaves in chemical reactions. Atoms contain protons, neutrons, and electrons, which influence an element’s reactivity, bonding, and state.
These properties occur at regular intervals across the periodic table, allowing chemists to classify elements into groups and periods.
What Is Atomic Structure?
Atoms are the building blocks of all matter. Understanding atomic structure is essential in chemistry, as it explains how elements react, form compounds, and determine the chemical properties of substances.
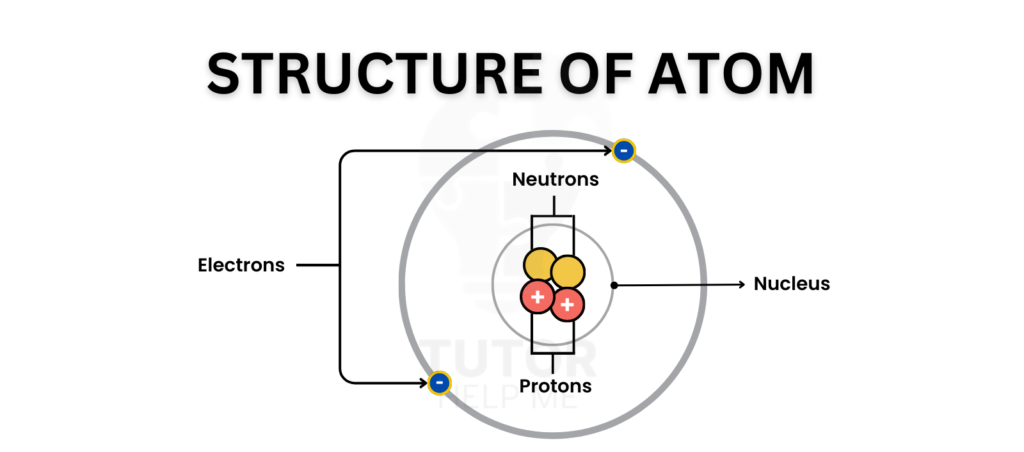
Every atom consists of a nucleus at its centre, surrounded by electrons arranged in shells. The nucleus contains protons and neutrons, while electrons orbit in energy levels around it.
Protons, Neutrons, and Electrons
Protons
- Charge: Positive charge (+1)
- Location: Found within the nucleus of an atom.
- Mass: 1 atomic mass unit (amu).
- Role: The number of protons in the nucleus determines the atomic number of an element. This number is unique for each element and defines its place in the periodic table.
Example: Hydrogen has 1 proton, while oxygen has 8 protons.
Neutrons
- Charge: No charge (neutral)
- Location: Found within the nucleus, alongside protons.
- Mass: 1 atomic mass unit (amu) (approximately the same as a proton).
- Role: Neutrons help stabilise the nucleus by reducing repulsion between positively charged protons. Elements can have different numbers of neutrons, creating isotopes.
Example: Carbon-12 has 6 neutrons, while Carbon-14 has 8 neutrons but remains the same element because it has 6 protons.
Electrons
- Charge: Negative charge (-1)
- Location: Orbiting the nucleus in electron shells.
- Mass: Very small (~1/1836 of a proton’s mass).
- Role: Electrons determine chemical properties and bonding. The number of electrons is equal to the number of protons in a neutral atom.
Example: Sodium has 11 protons and 11 electrons, making it electrically neutral.
"Get success in chemistry with our 1-on-1 GCSE chemistry tutor."
Mass and Charge of Subatomic Particles
The table below summarises the properties of protons, neutrons, and electrons:
| Subatomic Particle | Charge | Mass (amu) | Location in the Atom |
|---|---|---|---|
| Proton | +1 | 1 | Nucleus |
| Neutron | 0 | 1 | Nucleus |
| Electron | -1 | ~1/1836 | Orbiting the nucleus in shells |
Structure of an Atom
The Nucleus
The nucleus is found at the centre of an atom and contains protons and neutrons. It carries most of the mass of an atom, as electrons have very little mass.
The protons in the nucleus have a positive charge, while the neutrons are neutral, meaning the nucleus is positively charged overall.
Electron Shells and Energy Levels
Electrons move around the nucleus in specific regions called shells or energy levels.
- The first shell can hold up to 2 electrons.
- The second shell can hold up to 8 electrons.
- The third shell can hold up to 8 electrons.
The electronic structures of elements determine how they react. For example, elements in the same group in the periodic table have similar chemical properties because they have the same number of electrons in their outer shell.
How Are Electrons Arranged in an Atom?
Atoms contain protons, neutrons, and electrons, with electrons arranged in shells around the nucleus. Understanding how electrons are arranged helps explain chemical reactions, bonding, and the periodic table.
Electron Shells and Energy Levels
Electrons are found in energy levels (shells) surrounding the nucleus. These shells follow a specific order of atomic structure, with lower energy levels filled first.
- The first shell holds up to 2 electrons.
- The second shell holds up to 8 electrons.
- The third shell holds up to 8 electrons.
Electrons in their outer shell determine an element’s chemical properties.
How to Determine the Electronic Configuration of Elements
The electronic configuration of an atom shows how its electrons are arranged. It follows the pattern of filling electron shells in order of increasing energy levels.
Steps to determine electronic configuration:
- Find the atomic number of the element (equal to the number of protons and electrons).
- Distribute the electrons into shells, following the rule: 2, 8, 8.
- The outermost shell determines an element’s group in the periodic table.
Examples of Electronic Configuration for Common Elements
| Element | Atomic Number | Electronic Configuration |
|---|---|---|
| Hydrogen | 1 | 1 |
| Helium | 2 | 2 |
| Oxygen | 8 | 2,6 |
| Sodium | 11 | 2,8,1 |
| Chlorine | 17 | 2,8,7 |
| Calcium | 20 | 2,8,8,2 |
- Sodium (Na) has 1 outer electron, making it a Group 1 metal.
- Chlorine (Cl) has 7 outer electrons, placing it in Group 7 (halogens).
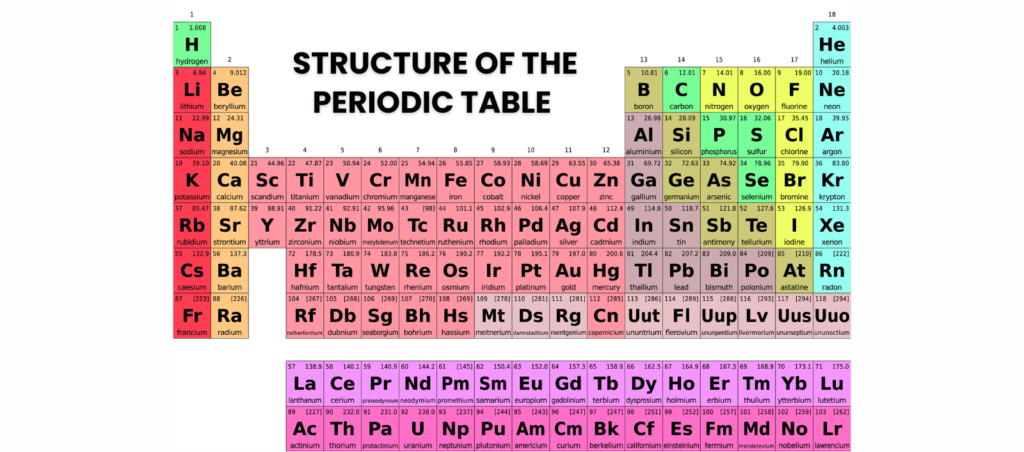
What Is the Periodic Table?
The periodic table is an organised chart of all known chemical elements, arranged by atomic number. It helps chemists predict chemical properties and reactions.
How Elements Are Organised by Atomic Number
The periodic table is structured by increasing atomic number, which represents the number of protons in the nucleus.
- Each element has a unique atomic number (e.g., Hydrogen = 1, Oxygen = 8).
- The relative atomic mass is also shown, which includes protons and neutrons.
- Elements in the periodic table are classified into metals, non-metals, and metalloids.
Periods (Rows) and Groups (Columns)
The modern periodic table is divided into periods (rows) and groups (columns).
Periods (Horizontal Rows)
- There are 7 periods in the periodic table.
- The period number represents the number of electron shells an atom has.
Groups (Vertical Columns)
- There are 18 groups, with elements in the same group having similar properties.
- Each group shares the same number of outer electrons, affecting chemical reactivity.
| Group | Example Elements | Key Properties |
|---|---|---|
| Group 1 (Alkali Metals) | Lithium, Sodium, Potassium | Very reactive, form alkaline solutions |
| Group 7 (Halogens) | Fluorine, Chlorine, Iodine | Form salts, react with metals |
| Group 8/0 (Noble Gases) | Helium, Neon, Argon | Unreactive, full outer shell |
Key Trends in the Periodic Table
- Reactivity increases down Group 1 but decreases down Group 7.
- Metals are on the left side, while non-metals are on the right.
- Transition metals (middle section) are used in catalysts and industry.
How Do Atomic Number and Mass Number Differ?
Atoms are the building blocks of all matter, and each element in the periodic table is defined by its atomic number and mass number.
These two numbers help chemists understand an element’s structure and properties.
Atomic Number
The atomic number of an element is the number of protons in the nucleus of an atom. It determines an element’s position in the periodic table and its chemical properties.
For example:
- Hydrogen has one proton, so its atomic number is 1.
- Oxygen has eight protons, so its atomic number is 8.
Since atoms are neutral, the number of protons is always equal to the number of electrons in an atom.
Mass Number
The mass number of an atom is the total number of protons and neutrons in the nucleus.
Formula:
Mass Number = Number of Protons + Number of Neutrons
For example:
- Carbon-12 has 6 protons and 6 neutrons, so its mass number is 12.
- Chlorine-35 has 17 protons and 18 neutrons, so its mass number is 35.
The mass of an atom depends on the number of protons and neutrons, as electrons have negligible mass.
How Do Isotopes Affect Atomic Mass?
Some elements have different versions of the same atom, called isotopes. Isotopes have the same atomic number (same number of protons) but different numbers of neutrons, leading to different mass numbers.
For example:
- Carbon-12 (¹²C) has 6 protons and 6 neutrons.
- Carbon-14 (¹⁴C) has 6 protons and 8 neutrons.
Since elements exist as a mixture of isotopes, their atomic mass is calculated as a relative atomic mass, which considers the average mass of all isotopes.
For chlorine:
Relative Atomic Mass= (35×75) + (37×25) / 100 = 35.5
This is why chlorine’s atomic mass on the periodic table is 35.5, even though no atom of chlorine actually has this exact mass.
What Are Isotopes?
Isotopes are atoms of the same element that have the same atomic number but different mass numbers due to a different number of neutrons.
Examples of Isotopes
| Element | Common Isotopes | Protons | Neutrons | Uses |
|---|---|---|---|---|
| Carbon | Carbon-12, Carbon-14 | 6 | 6 or 8 | Carbon-14 is used in radiocarbon dating |
| Chlorine | Chlorine-35, Chlorine-37 | 17 | 18 or 20 | Used in the chemical industry and disinfection |
| Iodine | Iodine-127, Iodine-131 | 53 | 74 or 78 | Iodine-131 is used in medical treatments |
What Are the Different Element Groups in the Periodic Table?
The periodic table arranges elements based on their atomic number and chemical properties. Elements within the same group in the periodic table share similar chemical properties because they have the same number of electrons in their outer shell.
Group 1 – Alkali Metals
The alkali metals are found in Group 1 of the periodic table. These elements are highly reactive metals because they have one electron in their outer shell.
Properties of Alkali Metals
- Soft and can be cut with a knife.
- Low density (e.g., lithium, sodium, and potassium float on water).
- React violently with water to form an alkaline solution.
- Stored in oil to prevent reaction with oxygen.
Examples of Alkali Metals
- Lithium (Li) – Used in batteries.
- Sodium (Na) – Found in table salt (sodium chloride).
- Potassium (K) – Essential for plant growth in fertilisers.
Group 2 – Alkaline Earth Metals
The alkaline earth metals are located in Group 2. They are less reactive than Group 1 metals but still react with water and acids.
Properties of Alkaline Earth Metals
- Harder and denser than alkali metals.
- React with water, though less violently than Group 1.
- Good conductors of electricity.
Examples of Alkaline Earth Metals
- Magnesium (Mg) – Used in aerospace materials and fireworks.
- Calcium (Ca) – Found in bones, teeth, and limestone.
Group 7 – Halogens
The halogens are found in Group 7 and are non-metals. The word ‘halogen’ means "salt former" because these elements react with metals to form salts.
Properties of Halogens
- Toxic and reactive.
- Exist as diatomic molecules (e.g., Cl₂, F₂, Br₂).
- Melting points and boiling points increase down the group.
Examples of Halogens
- Fluorine (F₂) – Found in toothpaste to prevent decay.
- Chlorine (Cl₂) – Used in swimming pools for disinfection.
- Iodine (I₂) – Essential for thyroid health.
Group 8/0 – Noble Gases
The noble gases in Group 8 (or Group 0) are chemically unreactive because they have a full outer electron shell.
Properties of Noble Gases
- Colourless, odourless gases.
- Low boiling points.
- Do not easily form compounds.
Examples of Noble Gases
- Helium (He) – Used in balloons and airships.
- Neon (Ne) – Found in neon signs.
- Argon (Ar) – Used in light bulbs to prevent filament burning.
Transition Metals
The transition metals are located in the middle block of the periodic table. These metals have unique chemical properties and are commonly used in construction and industry.
Properties of Transition Metals
- Good conductors of heat and electricity.
- High melting points.
- Form coloured compounds (e.g., copper sulfate is blue).
- Used as catalysts in chemical reactions.
Examples of Transition Metals
- Iron (Fe) – Used in construction and steel production.
- Copper (Cu) – Found in wires and plumbing.
- Gold (Au) – Used in jewellery and electronics.
How Do Metals and Non-Metals Differ?
The periodic table is divided into metals and non-metals, which have opposite properties. The metalloids (found along the “stair-step” line) share characteristics of both.
Characteristics of Metals
- Good conductors of electricity and heat.
- Malleable (can be shaped into thin sheets).
- Ductile (can be drawn into wires).
- High melting and boiling points.
- Shiny and solid at room temperature (except mercury).
Examples of Metals
- Aluminium (Al) – Used in aeroplanes.
- Copper (Cu) – Used in electrical wiring.
Characteristics of Non-Metals
- Poor conductors of electricity and heat.
- Brittle (breaks easily).
- Low boiling and melting points.
- Can exist as solids, liquids, or gases at room temperature.
Examples of Non-Metals
- Oxygen (O₂) – Essential for respiration.
- Sulphur (S) – Used in fertilisers and gunpowder.
What Are Metalloids?
Metalloids have properties of both metals and non-metals. They are useful in electronics and semiconductors.
Examples of Metalloids
- Silicon (Si) – Used in computer chips.
- Boron (B) – Found in glass and detergents.
Read more What Are Acids, Bases, and Salts?
FAQ’s
What is the smallest particle of an element?
An atom is the smallest particle of an element, retaining its chemical properties and consisting of protons, neutrons, and electrons.
Why do elements in the same group have similar properties?
Elements in the same group have similar properties due to having the same number of valence electrons, affecting their chemical behavior.
How do scientists discover new elements?
Scientists discover new elements through nuclear reactions, particle accelerators, and experiments that create and identify previously unknown atomic structures.
Why is the periodic table important in chemistry?
The periodic table organizes elements, predicting properties, trends, and reactivity, making it essential for chemistry studies and scientific research.